Chengdu, August 15, 2025 — Chengdu Hyperway Pharmaceutical Co., Ltd. ("Hyperway Pharma") has officially submitted an Investigational New Drug (IND) application to CDE for its novel oral Pan-KRAS inhibitor, HBW-012462. This submission marks a major milestone in Hyperway Pharma’s pursuit of innovative oncology therapeutics, targeting KRAS—a historically “undruggable” oncogene implicated in some of the most treatment-resistant cancers.
In parallel, Hyperway Pharma has initiated a Phase II clinical trial application with the U.S. FDA for its flagship analgesic candidate, HBW-004285 tablets, signaling the company’s formal entry into global clinical development.
KRAS Mutations: A High-Impact Target in Oncology
KRAS mutations are present in approximately 30% of all cancers, with seven dominant subtypes accounting for over 90% of cases. These mutations are especially predominant in hard-to-treat malignancies
Cancer Type | KRAS Mutation Rate | Common Subtypes |
Pancreatic Cancer | 86% | G12D, G12V, G12R |
Colorectal Cancer | 41% | G12D, G12V, G13X |
Lung Adenocarcinoma | 32% | G12C, G12V, G12D |
Pan-KRAS inhibitors, which target multiple KRAS mutations simultaneously, represent a transformative approach in oncology. Unlike single-mutation inhibitors, they offer broader applicability, potential to overcome resistance, and enhanced synergy with other therapies. Analysts estimate the market for KRAS G12C and G12D inhibitors alone could exceed $10 billion, with Pan-KRAS inhibitors projected to reach up to eight times that value.
HBW-012462: A Next-Generation Pan-KRAS Inhibitor
Hyperway Pharma’s HBW-012462 is a potent, selective, and tissue-enriched oral Pan-KRAS inhibitor capable of targeting both KRAS (ON) and KRAS (OFF) states. Compared to publicly available data on Eli Lilly’s LY4066434 and Pfizer’s PF-07934040, HBW-012462 demonstrates competitive or superior performance across key pharmacological metrics.
Key Development Highlights:
Potency & Selectivity: Inhibits proliferation of KRAS-mutant cell lines (GP2D, RKN, PANC-1, NCI-H1373) with no activity against HRAS, NRAS, or wild-type KRAS.
Tissue Enrichment: Exhibits 10–100x higher exposure in target organs (pancreas, colon, lung) vs. plasma in preclinical models.
Anti-Tumor Efficacy: Demonstrates robust tumor shrinkage and elimination across KRAS-mutant mouse models, with clear dose-response and tolerability.
Synergistic Potential: Combination with FAK or EGFR inhibitors leads to complete tumor suppression and regression.
Safety Profile: 28-day GLP toxicology studies in rats and dogs indicate a safety margin exceeding 15x, supporting clinical advancement.
Preclinical data were showcased at the 2025 AACR Annual Meeting, reinforcing HBW-012462’s promise as a next-generation therapeutic in KRAS-driven cancers.
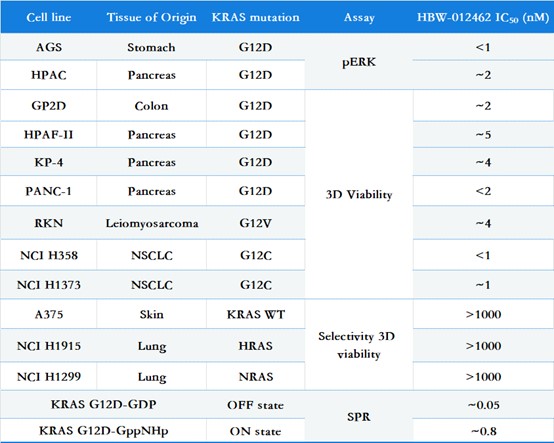
(Note: Partial biological activity data of preclinical HBW-012462)
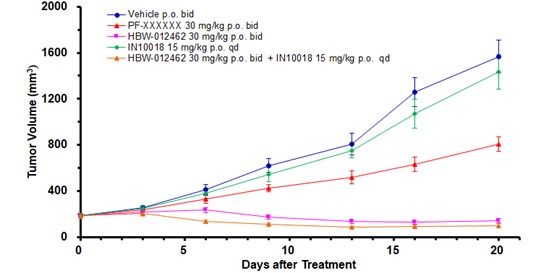
(IN10018 is a FAK inhibitor)
(Note: Drug combination study in NCI-H1373 KRAS G12C (NSCLC) mouse model)

Contact:Miss Jiang
Telephone:028-87014968
Email:xiameijiang@hyperwaypharma.com
Address:8Th Floor, Building B4, Tianfu Life Science Park, No.88, Keyuan South Road, Hi-Tech Zone, Chengdu, Sichuan